Introduction : Matter Transformation
Hey, future chemists and master builders! Ever stopped to think why everything around you can change? Like, how does a yummy cake turn into energy for your body? Or how can boring old elements become something as amazing as YOU? This is all thanks to the secret language of Matter Transformation!
You know how physics (think Newton and falling apples) taught us that the universe moves in super predictable ways? But just knowing how things move isn’t the whole story. Motion alone can’t explain why:
- Iron gets rusty (that’s a cool Matter Transformation!)
- Wood burns into ash and smoke
- Invisible gases like hydrogen and oxygen suddenly become water you can drink!
- Simple stuff combines to make something as complex as a human being!
That’s where chemistry steps in! If physics tells us how things move, chemistry reveals how things become – how they change, mix, and unlock completely new possibilities. It’s like discovering the universe’s secret code for building and changing everything through Matter Transformation!

Various atoms bonding and together, showcasing the fundamental process of Matter Transformation.
Alchemy trying for Matter Transformation
Long, long ago, before chemistry was a proper science, people called alchemists were the original mad scientists! They weren’t just trying to turn lead into gold (though that was a popular dream!). Across ancient cultures, they were heating, dissolving, mixing, and distilling all sorts of substances, fascinated by Matter Transformation.

When Did Alchemy End for Matter Transformation
They were part mystical magicians, part hands-on experimenters. Their goals were often a bit wild, but their core belief was spot-on: matter can change form, and these changes follow patterns. Their busy hands, even with all the mysterious symbols, were paving the way for real chemical discovery and understanding Matter Transformation!
Lavoisier: The Chemist Who Proved Nothing Disappears During Matter Transformation!

The big step from magical alchemy to real chemistry happened when people started getting super precise. Meet Antoine Lavoisier, the brilliant mind who brought careful measurement into the lab.
He performed groundbreaking experiments that showed something amazing: matter is neither created nor destroyed in a chemical reaction; it just transforms! This is the ultimate rule of Matter Transformation! When wood burns, it doesn’t vanish; it becomes ash, smoke, and gases, and all those parts still weigh the same as the original wood!
This idea, called the Conservation of Mass, totally stripped away the magic from reactions. They weren’t mysterious events; they were just clever rearrangements of matter! Suddenly, curiosity had numbers and facts to back it up. Substances weren’t mysterious spirits; they were measurable combinations of elements constantly undergoing Matter Transformation.
Mendeleev: The Guy Who Predicted the Future

Then came Dmitri Mendeleev, who looked at all the known chemical elements and noticed something incredible – a pattern! He wasn’t just making a list; he was arranging them by their properties and atomic weight, and he saw a repeating rhythm. He created the Periodic Table, which you probably know today!
The most astonishing part? He left gaps in his table and predicted elements that hadn’t even been discovered yet! And guess what? Years later, those elements were found, exactly where he said they’d be and with the properties he described! Mendeleev’s table showed us that nature’s incredible diversity comes from an underlying, beautiful order, guiding every Matter Transformation. It was another hint that beneath all the different “stuff,” there was a deeper unity.
The Invisible Bonds: How Things Connect Matter
As chemistry got even deeper, scientists realized something mind-blowing: matter isn’t just a bunch of individual particles. It’s all about relationships! Atoms don’t just exist; they bond with each other. They share electrons, they transfer them, and invisible forces hold these new structures together. This is the heart of Matter Transformation!
Chemistry showed us that transformation depends on connection.
- Hydrogen and oxygen, two invisible gases, combine to make water – a totally different liquid with new properties!
- Carbon, the ultimate team player, bonds in endless ways to form all the organic molecules that make up life itself!
This isn’t just about things moving around; it’s about creative combination! Simple building blocks, through their amazing connections, create all the incredible complexity we see in the world through Matter Transformation.
Chemistry: The Bridge to Life and Endless Matter Transformation!
This is where chemistry gets super exciting – it’s the bridge between the physics of motion and the biology of life! Without chemical bonds, there’s no metabolism (how your body uses energy). Without complex molecules, there are no cells. The laws of motion explain how particles move. Chemistry explains how they organize themselves into something more. And in that organization lies the very spark of life, driven by constant Matter Transformation!
From the versatility of carbon forming endless molecules to DNA encoding information in chemical sequences, your very existence is a chemical miracle! The lab bench, where chemists mix and match, becomes the threshold where matter starts hinting at life and its amazing Matter Transformation.
Matter Transformation Is the Universal Rule!
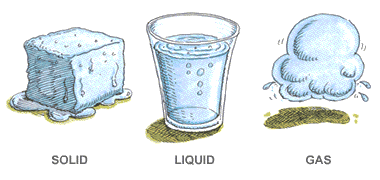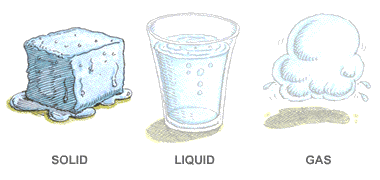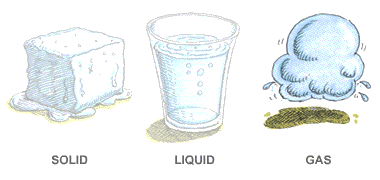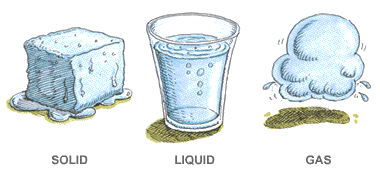
Chemistry teaches us a huge lesson: things aren’t fixed! They can always transform without breaking any rules.
- Water can be ice, liquid, or vapor.
- Iron can rust.
- Carbon can be a soft pencil lead or a super-hard diamond!
The world isn’t static; it’s always changing! If physics showed us that motion follows laws, chemistry reveals that Matter Transformation also follows laws. Change isn’t chaos; it’s just another form of order.
What’s Next? Beyond the Bonds of Matter Transformation!
Chemistry has given us so much – new medicines, amazing materials, fertilizers to feed the world! But curiosity never stops!
If atoms combine using invisible forces, what are atoms themselves? What’s inside them? Why do these bonds form in the exact ways they do?
The answers to these questions would require an even bigger revolution – a journey into the quantum realm, where the clear-cut rules of physics and chemistry start to get blurry, and probability takes over.
Chemistry brought us to the very edge of the invisible. To go further, we’d have to rethink matter itself, diving from the bonds of chemicals to the mysteries of subatomic particles.
From predictable Matter Transformation to mind-bending uncertainty. From solid bonds to wavy fields. From laboratory glassware to subatomic mystery! And that’s where our next adventure awaits!
Click here for our next Journey: Invisible Worlds: Atoms, Energy, and the Quantum Turn!